-
Choosing the Right Enzyme Immobilization Chemistry: A Strategic Guide for Scalable Biocatalysis
Selecting the correct enzyme immobilization chemistry is critical for process stability and cost. This guide compares covalent, metal affinity, ionic, and adsorption methods to help PIs and Innovators optimize biocatalyst performance for industrial scale-up. 💬 Need help? Talk to a Solidzymes expert today. Introduction: Why Enzyme Immobilization Chemistry Choice is A Crucial Scale-Up Decision In

|
-
Controlling the Reaction: A Practical Guide to Enzyme Kinetics & Active Sites
For the industrial scientist, speed in enzyme kinetics isn’t just a metric; it is the difference between a bench-scale curiosity and a commercially viable process. While the fundamental definition of a catalyst is elementary, the engineering required to optimize that catalyst for specific industrial conditions is anything but. In the realm of enzyme kinetics, we

|
-
The PI’s Guide to E. Coli Expression: From Sequence to High-Yield Sample
In research, every second and every microgram of protein counts. Grant deadlines loom, publications are pending, and you need a reliable, cost-effective, and scalable method to produce your protein of interest. While newer, more complex expression systems get a lot of attention, one host remains the undisputed workhorse of molecular biology: Escherichia coli. For Principal

|
-
Purifying an Enzyme or Scaling a Biocatalyst? Choosing the Right Immobilization Strategy
You’ve done it. After countless hours in the lab, you’ve engineered the perfect enzyme. It has the potential to revolutionize a manufacturing process or unlock a new frontier in your research. But now comes the critical next step: your immobilization strategy. Attaching your protein to a solid surface is essential, but it’s not a one-size-fits-all

|
-
Lactase Case Study
The beta-galactosidase enzyme, also known as lactase, is an industrial biocatalyst used to produce lactose free milk (Horner, 2011). The activity of this enzyme is easy to measure and it is commercially available (Biosynth FL31718) making it an excellent model to test Solidzymes’ carrier screening methodology and equipment. Enzyme carriers were first assessed for their

|
-
Co-Immobilization of Enzymes: The Innovation Reshaping Bioprocessing
Why Are We Excited About Co-Immobilization of Enzymes? If you’re working in food tech, biopharma, pharma process chemistry, or procurement, you know the challenges: long development cycles, high enzyme costs, unstable intermediates, and scalability hurdles. One strategy consistently changing the game? Co-immobilizing multiple enzymes on a single carrier, enabling them to act in sequence within

|
-
Maximizing cost efficiency through enzyme immobilization
The need for affordable and sustainable approaches within industrial biotechnology continues to grow. One of the most revolutionary breakthroughs has been industrial enzyme immobilization. By adsorbing enzymes onto solid supports this technique boosts enzyme stability, allows enzyme reuse in the industry, and facilitates re-use in multiple reaction cycles. In addition, reducing the cost of goods

|
-
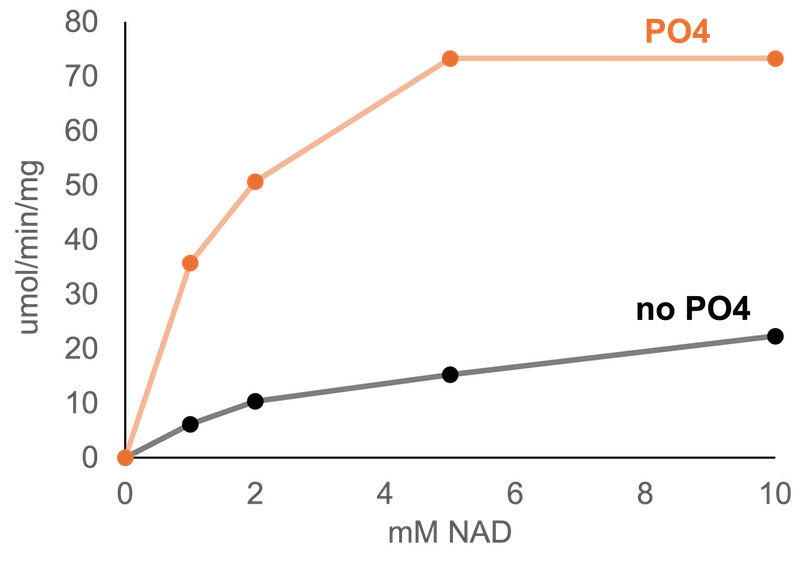
Glucose-6-phosphate Dehydrogenase characterization
Expression of His tagged proteins in E. Coli followed by purification using nickel agarose beads is the method of choice to produce most proteins at Solidzymes. This is probably the most common method to produce recombinant proteins at lab scale, however, each lab does it a little differently. In this post we describe purification of

|
-
Lactase Stabilization with Glutaraldehyde
Glutaraldehyde is a well known covalent crosslinker that is often used to improve the enzyme immobilization process by creating covalent attachment points (Rodrigues et al. 2021). It reacts with amine groups on the surface of proteins / particles and in so doing promotes structural integrity of the enzyme by connecting it to the solid support,

|
Insights & Innovations
Here you can read about current events and examples from finished projects.